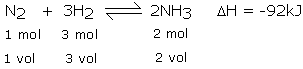Production of Ammonia through the Haber-Bosch Process
When combined together, gaseous hydrogen and nitrogen gives ammonia. Nitrogen from the air and hydrogen from natural gases (mainly methane) and the gaseous ammonia is cooled down and liquiefied. However, the process is not as easy as it sounds. As the above reaction is reversible, the ammonia formed can easily change back into nitrogen and hydrogen gas. In order for ammonia to be produced at an optimal rate on a large scale, several conditions must be met.
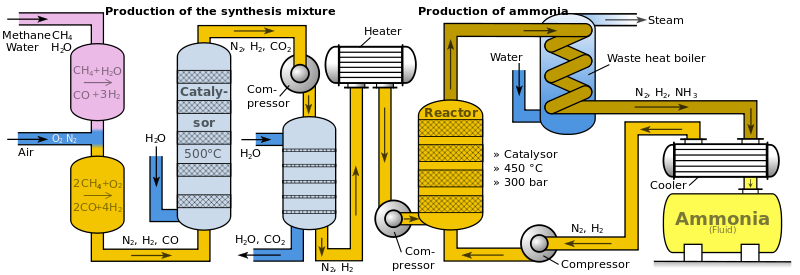
Basically, in simple steps:
1) Hydrogen and Nitrogen are extracted and purified with all impurities removed for the process.
2) Both gases are mixed, and the mixture is pumped into a compressor. The compressor has extreme conditions, with a pressure of 200atm. The high pressure causes the particles to gain kinetic energy and makes the temperature rise up to about 300 degrees Celsius. This causes the covalent bonds in the hydrogen and nitrogen molecules to split up.
3) The compressed molecules now enter a converter. The converter is a tank heated up to a temperature of 400-450 degrees Celsius, and is pressurized at 200atm. Inside the compressor, there are shelves of hot iron (Fe3+) to act as the catalyst. Aluminum oxide (Al2O3) and potassium oxide (K2O) are used as promoters to increase the catalyst's efficiency.
4) The gaseous hydrogen and nitrogen reacts on the surface of the hot iron to form gaseous ammonia. However, some of the gases remain unreacted. In addition to the above, the production of ammonia is a reversible reaction, and so some of the ammonia formed will decompose back to hydrogen and nitrogen gas. As a result, the final yield is only about 12-15% of its original reactants.
5) The mixture of hydrogen, nitrogen and ammonia is then removed and cooled, where under the right pressure, the ammonia liquiefies and is collected.
6) The unreacted hydrogen and nitrogen gases is sent back into the converter, and steps 3, 4 and 5 are repeated.
Basically, in simple steps:
1) Hydrogen and Nitrogen are extracted and purified with all impurities removed for the process.
2) Both gases are mixed, and the mixture is pumped into a compressor. The compressor has extreme conditions, with a pressure of 200atm. The high pressure causes the particles to gain kinetic energy and makes the temperature rise up to about 300 degrees Celsius. This causes the covalent bonds in the hydrogen and nitrogen molecules to split up.
3) The compressed molecules now enter a converter. The converter is a tank heated up to a temperature of 400-450 degrees Celsius, and is pressurized at 200atm. Inside the compressor, there are shelves of hot iron (Fe3+) to act as the catalyst. Aluminum oxide (Al2O3) and potassium oxide (K2O) are used as promoters to increase the catalyst's efficiency.
4) The gaseous hydrogen and nitrogen reacts on the surface of the hot iron to form gaseous ammonia. However, some of the gases remain unreacted. In addition to the above, the production of ammonia is a reversible reaction, and so some of the ammonia formed will decompose back to hydrogen and nitrogen gas. As a result, the final yield is only about 12-15% of its original reactants.
5) The mixture of hydrogen, nitrogen and ammonia is then removed and cooled, where under the right pressure, the ammonia liquiefies and is collected.
6) The unreacted hydrogen and nitrogen gases is sent back into the converter, and steps 3, 4 and 5 are repeated.
Why these conditions?
As the production of ammonia is a reversible reaction, where the forward reaction is what we want to obtain the final product ammonia, according to Le Chatelier's Principle, the best conditions to get the most products out of the reaction would be low temperatures and high pressures. However, as seen from the process above, 400-450 degrees Celsius is nowhere near low and 200atm is not an extremely high pressure. This is because there are many other conditions to take into consideration.
Keeping Le Chatelier's Principle in mind:
Any change in a system at equilibrium results in a shift of the equilibrium in the direction which minimises the change.
Keeping Le Chatelier's Principle in mind:
Any change in a system at equilibrium results in a shift of the equilibrium in the direction which minimises the change.
The temperature:
As seen above, the forward reaction is exothermic, which would mean that the lower the temperature, the more the equilibrium would shift to the left to produce more heat, giving us more of the product.
However, at low temperatures, the yield produced would be very minimal in a short time due to the lack of kinetic energy the particles would have. With low kinetic energy, the particles would react very slowly. Ammonia manufacturers need to produce a large amount of ammonia per day in order to make the business a profitable one. Henceforth, 400-450 degrees Celsius is a compromise temperature set to produce a relatively high amount of ammonia in a very short time.
The pressure:
In the equation above, it can be seen that there are 4 mols of gaseous molecules on the left and only 2 mols of gaseous molecules on the right. According to Le Chatelier's Principle, increasing the pressure would shift the equilibrium to the right, favouring the forward reaction and producing more ammonia. The high pressure would bring the molecules closer together, increasing the chances of them colliding and reacting to form ammonia.
However, to achieve high pressures, a lot of money is needed to build and maintain the pipes and machines. Strong pipes and tanks will need to be built in order to withstand the high pressure and yet work efficiently. The total running costs of ammonia production factories at very high pressures will be very expensive. Henceforth, 200atm is a compromise pressure set to maximise profits.
As seen above, the forward reaction is exothermic, which would mean that the lower the temperature, the more the equilibrium would shift to the left to produce more heat, giving us more of the product.
However, at low temperatures, the yield produced would be very minimal in a short time due to the lack of kinetic energy the particles would have. With low kinetic energy, the particles would react very slowly. Ammonia manufacturers need to produce a large amount of ammonia per day in order to make the business a profitable one. Henceforth, 400-450 degrees Celsius is a compromise temperature set to produce a relatively high amount of ammonia in a very short time.
The pressure:
In the equation above, it can be seen that there are 4 mols of gaseous molecules on the left and only 2 mols of gaseous molecules on the right. According to Le Chatelier's Principle, increasing the pressure would shift the equilibrium to the right, favouring the forward reaction and producing more ammonia. The high pressure would bring the molecules closer together, increasing the chances of them colliding and reacting to form ammonia.
However, to achieve high pressures, a lot of money is needed to build and maintain the pipes and machines. Strong pipes and tanks will need to be built in order to withstand the high pressure and yet work efficiently. The total running costs of ammonia production factories at very high pressures will be very expensive. Henceforth, 200atm is a compromise pressure set to maximise profits.